Ernest Rutherford
Rutherford changed the atomic model in 1910 from the previous Thomson model.
What did he do to change the atomic model?
He discovered the NUCLEUS!
The Gold Foil Experiment
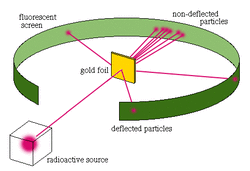
Rutherford wanted to investigate the structure of atoms. He devised the experiment shown here.
In this experiment, alpha particles were shot at a thin sheet made of gold atoms. A detector screen surrounding the foil would light up every time it was hit by an alpha particle so Rutherford could see where the alpha particles ended up.
In this experiment, alpha particles were shot at a thin sheet made of gold atoms. A detector screen surrounding the foil would light up every time it was hit by an alpha particle so Rutherford could see where the alpha particles ended up.
Picture Source: http://3.bp.blogspot.com/-HKmSsop67RU/TXLKsObCsBI/AAAAAAAABso/3YQTbyT9AwI/s1600/atomErnest_Rutherford_2.gif
Here's an animation that shows how his experiment worked
What happened?
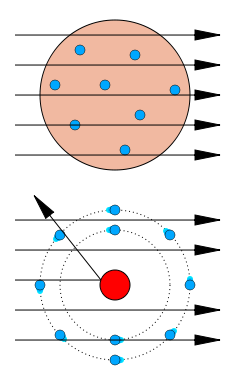
You can imagine his surprise when some alpha particles started bouncing back in his direction!
Based on the Thomson model, he expected the particles to pass right through the atoms. (Top right)
However, here were alpha particles bouncing off something within the atoms! (Bottom right image)
What did this mean?
Rutherford concluded that there was a positively charged nucleus. He also concluded that atoms are mostly empty space and that the electrons surrounded the positive nucleus.
Based on the Thomson model, he expected the particles to pass right through the atoms. (Top right)
However, here were alpha particles bouncing off something within the atoms! (Bottom right image)
What did this mean?
Rutherford concluded that there was a positively charged nucleus. He also concluded that atoms are mostly empty space and that the electrons surrounded the positive nucleus.
Picture Source: http://upload.wikimedia.org/wikipedia/commons/thumb/9/9b/Gold_foil_experiment_conclusions.svg/230px-Gold_foil_experiment_conclusions.svg.png
So what happened then?
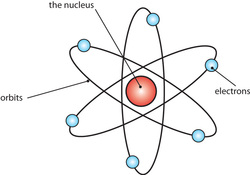
His discoveries led to this new model.
Picture Source: http://atomic.lindahall.org/siteart/questions/rutherford-atom-for-carbon_lg.jpg
