The Quantum Model
The quantum model is a model of the atom that is based on mathematics. This model is based on quantum theory. The quantum model uses probability to locate electrons by describing an orbital where an electron is likely to be found.
In other words, the model can't predict the exact location of an electron but is still very useful for observing atoms.
In other words, the model can't predict the exact location of an electron but is still very useful for observing atoms.
So, how do we use it?
Quantum Numbers
Well first you have to know about quantum numbers. These numbers help describe electrons and their orbitals.
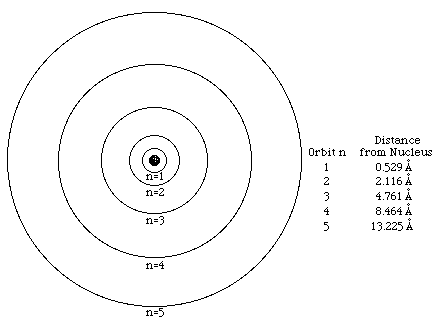
The Bohr model is only in 2 dimensions and only used one quantum number,
n
to talk about the size of the specific orbit of an electron.
The Bohr model is only in 2 dimensions and only used one quantum number,
n
to talk about the size of the specific orbit of an electron.
However...
The quantum model uses three dimensions! Therefore, scientists need more quantum numbers to describe it.
There are four quantum numbers used to describe electrons and their orbitals.
There are four quantum numbers used to describe electrons and their orbitals.
1-Principal Quantum Number: n
The principal quantum number is used to describe the distance of the orbital from the nucleus. It also describes the energy of the electron and the size of an orbital.
The principal quantum number can have positive values n=1, 2, 3, 4......... and so on.
The larger n gets, the larger the orbital is.
The principal quantum number can have positive values n=1, 2, 3, 4......... and so on.
The larger n gets, the larger the orbital is.
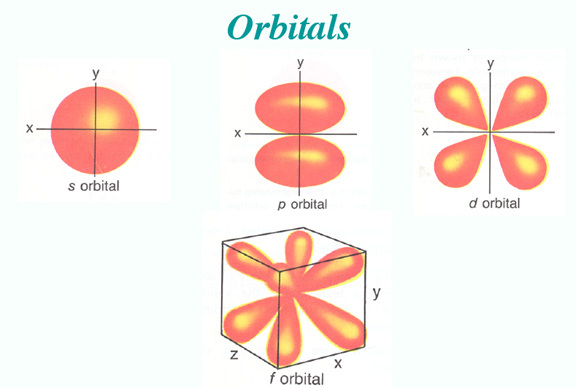
2-Angular Momentum Quantum Number: l
The angular momentum quantum number describes the shape of an orbital, as oppose to n describing the size.
l can have any value from 0 to (n-1). If an orbital has an n value of 2, then l=0, or 1.
l can have any value from 0 to (n-1). If an orbital has an n value of 2, then l=0, or 1.
What happens if orbitals have different l values but the same n value?
Scientists call these subshells. They name these subshells according to the l value:
These orbitals have different shapes. Orbital s has a sphere shape, orbital p has a polar shape, and orbital d has a cloverleaf shape. The shapes get more complex from there.
- 0 = s
- 1 = p
- 2 = d
- 3 = f
- 4 = g
These orbitals have different shapes. Orbital s has a sphere shape, orbital p has a polar shape, and orbital d has a cloverleaf shape. The shapes get more complex from there.
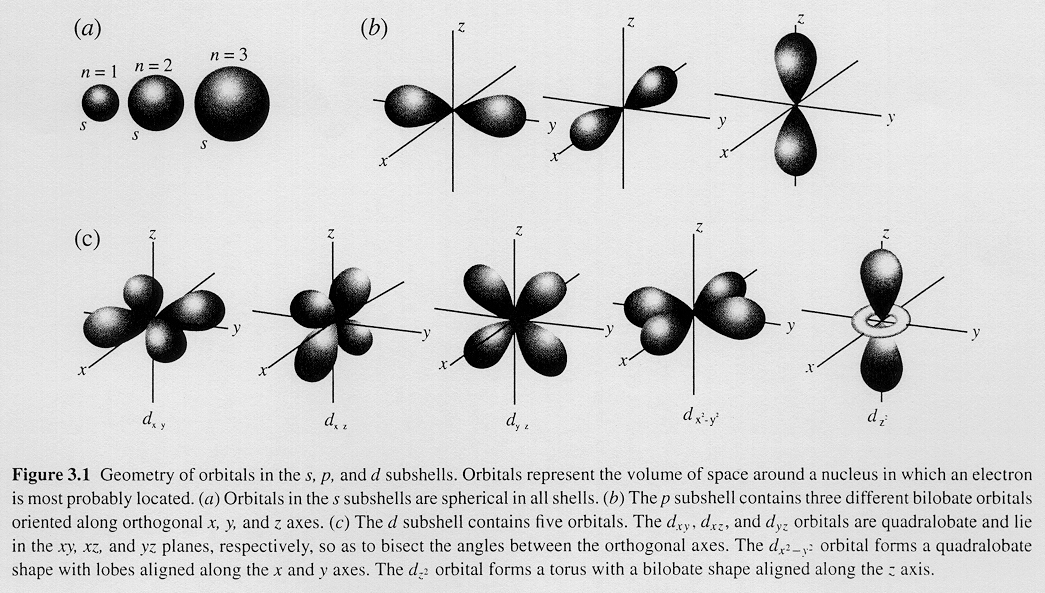
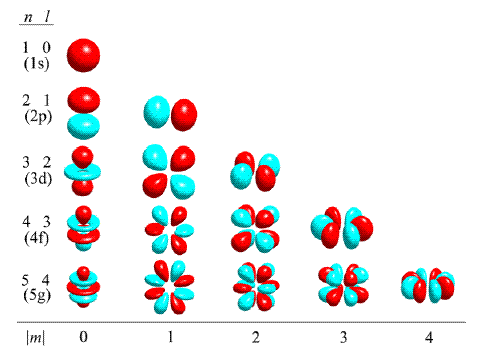
3- Magnetic Quantum Number: m
The magnetic quantum number describes the orientation of an orbital in space.
It can have values from -l to 0 to +l.
The drawing below shows how the value of m changes the way the orbitals are oriented.
It can have values from -l to 0 to +l.
The drawing below shows how the value of m changes the way the orbitals are oriented.
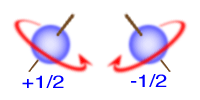
4- Spin Quantum Number
The spin quantum number describes whether an electron spins clockwise or counterclockwise in a magnetic field.
It can only have values of -1/2 or +1/2.
It can only have values of -1/2 or +1/2.