J.J. Thomson
J.J. Thompson was one of the pioneers in understanding atomic structure and constructing a more accurate model.
Well, what did he do exactly?
He discovered 'corpuscles', or what we call ELECTRONS!
But how did he achieve this?
Well, let's find out!
The Cathode Ray Experiment
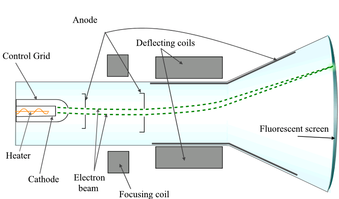
Thomson constructed an experiment like the one on the left. Basically, he was able to measure the ratio of the electrical charge of particles in a tube to their mass. He found that all gases contained the same particles, which are smaller than atoms.
These are what we now call electrons.
These are what we now call electrons.
Picture Source: http://creating.pbworks.com/f/Cathode_ray_Tube.PNG.png
This video shows how this experiment worked
What do electrons have to do with constructing a better atomic model?
The "Plum-Pudding" Model
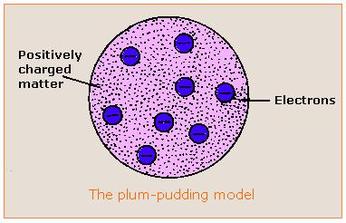
Well, since he had found electrons, Thomson was able to propose a new model!
He theorized that negatively-charged electrons were "floating around" in a "pudding" of positively-charged material.
This was radically different from the previously accepted Dalton model and changed the way science viewed the atom.
He theorized that negatively-charged electrons were "floating around" in a "pudding" of positively-charged material.
This was radically different from the previously accepted Dalton model and changed the way science viewed the atom.
Picture Source:http://images.tutorvista.com/content/atoms-and-nuclei/plum-pudding-model.jpeg
